steric hindrance
Steric hindrance
Steric hindrance is the slowing of chemical reactions due to steric bulk. It is usually manifested in intermolecular reactions, whereas discussion of steric effects often focus on intramolecular interactions. To put it simply, steric hindrance at a given atom in a molecule is the congestion caused by the physical presence of the surrounding ligands, which may slow down or prevent reactions at the atom.

In the second case, the carbonyl carbon is bonded to a hydrogen atom and a methyl group. Since the methyl group is larger than the hydrogen atom, steric hindrance is greater at the carbonyl carbon in 2 than that in 1.
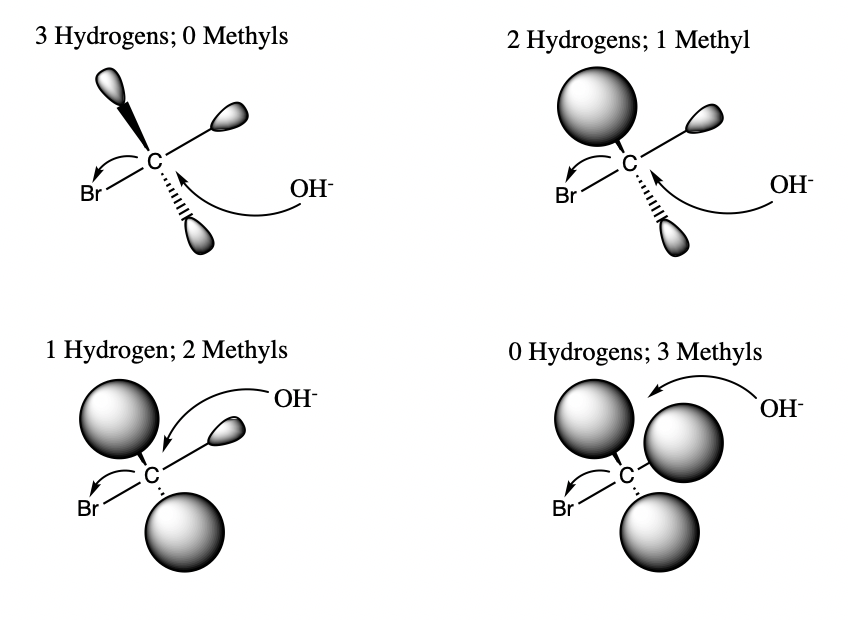
Here's another example. As more methyl groups are added to the molecule, there is less space for the covalent bond to the electrophile to form. Therefore, as the steric bulk increases, a molecule can be hindered from performing different reactions.