octet rule
Octet rule
The octet rule is a foundational principle in chemistry that explains how atoms form compounds to achieve a configuration akin to the noble gases, characterized by having eight electrons in their valence shells. The octet rule posits that atoms tend to engage in chemical reactions that allow them to have eight electrons in their valence shell. This configuration is deemed energetically favorable and mimics the electron configuration of noble gases, which are known for their chemical inertness and stablility.
Atoms with only one or two electrons in their outermost shell, such as sodium, tend to lose these electrons to form cations. This process is energetically favorable when the loss of electrons (an endothermic process) can be offset by subsequent electrostatic interactions in an ionic compound. Non-metals with six or seven valence electrons, such as fluorine or oxygen, can gain electrons to complete their octet. This process can be energetically favorable if the gain in stability from forming a full octet outweighs the energy required to add electrons to the existing shell.
Quote:
Regarding the octet concept, it's only a guideline at best, and nothing more. I wouldn't put too much stock into the concept. If you insist on using it, then only apply it to the following elements: C,N,O, and F.
Energetically favourable
- In full shells, the electron density is distributed evenly around the nucleus. This symmetric distribution reduces electron-electron repulsive forces because the electrons are spaced as far apart as possible within the constraints of their orbital shapes. For instance, in a p orbital set (\[p_{x}\], \[p_{y}\], \[p_{z}\]), having one electron in each or two electrons with opposite spins in each results in a balanced, symmetric spread of electron density.
The reason a fully filled block reduces the electron-electron repulsive force, despite having more electrons, is that when orbitals are fully occupied, the electron density is symmetrically distributed around the nucleus, which can minimize the overall electron-electron repulsion within that space. When orbitals are only partially filled, there is still potential for further electron arrangement that could lower the energy. Electrons in partially filled orbitals are more susceptible to repulsive interactions because they have fewer opportunities for beneficial symmetrical arrangements and exchange interactions.
In partially filled orbitals, not all available spatial configurations (orbitals) are maximally utilized. This can lead to a situation where electrons are more concentrated in fewer orbitals or regions of space, thereby increasing the electrostatic repulsion between them. Electrons repel each other, and having more electrons in closer proximity (as in the case of partially filled orbitals with multiple electrons) increases the repulsive force, which contributes to a higher total energy state of the system.
- The shape and phase of atomic orbitals lead to specific spatial distributions of electrons. Full s orbitals (spherical) and p orbitals (lobed in orthogonal directions) in their full configurations enhance this symmetry, contributing to stability by minimizing potential energy due to repulsions.
Full orbitals contribute to a symmetric electron distribution around the nucleus. Symmetry in the electron cloud minimizes the overall potential energy of the system by evenly distributing electrons and reducing focal points of negative charge concentration that increase repulsion. Partially filled orbitals do not contribute to such symmetry and can result in uneven distribution of electron density, which is energetically less favourable.
Exceptions
Elements in Period 3 and above that form covalent bonds can in fact not obey the octet rule if the resulting compound is sufficiently stable. An example of such a compound is \[\ce{SF6}\].
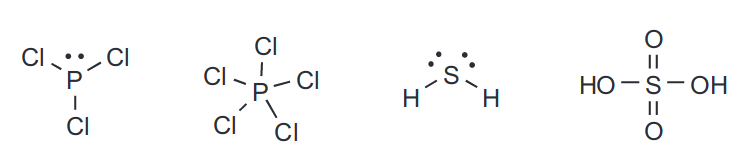
This is because the presence of d-orbitals which can accommodate up to ten electrons makes this possible.