electron configuration
Electron configuration
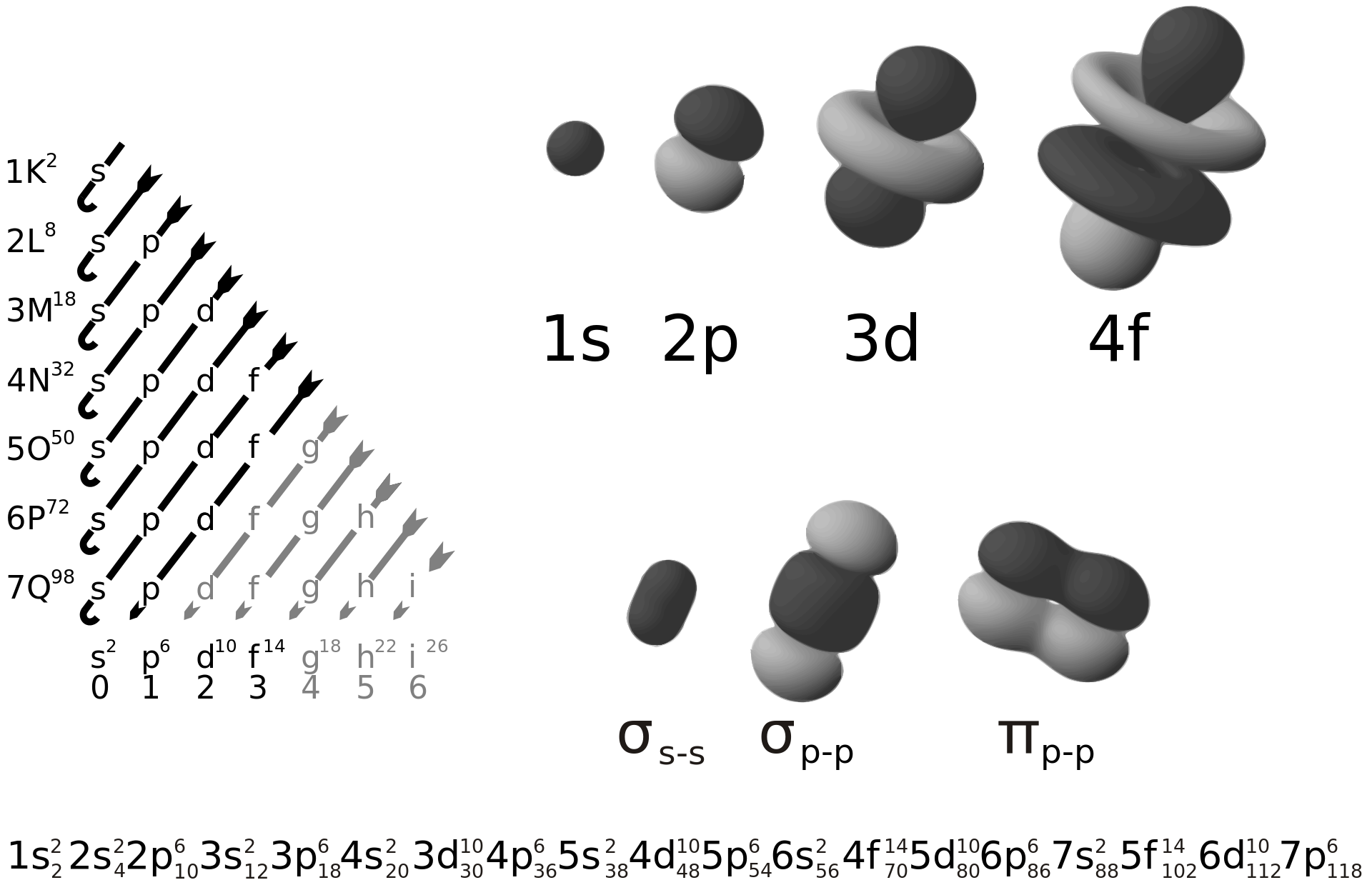
Electrons in atoms have only certain specific energy levels which are quantized (fixed). Electrons are organised according to their energy levels into shells, labelled by the principle quantum number.
Example:
The electron configuration for He (2 electrons) would be 1s2 and for Al (13 electrons) would be 1s22s22s63s23p1 or [Ne] 3s23p1 as the electron configuration for Ne is 1s22s22s6.
The electron configuration for Cl- would be [Ne] 3s23p6 while for Na+ it would be [He] 2s22p6.