emission spectrum
Emission spectrum
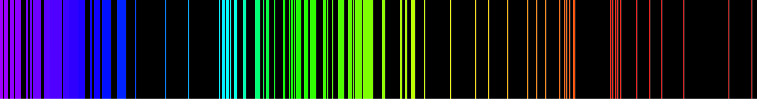
The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted due to electrons making a transition from a high energy state to a lower energy state. The photon energy of the emitted photons is equal to the energy difference between the two states. There are many possible electron transitions for each atom, and each transition has a specific energy difference. This collection of different transitions, leading to different radiated wavelengths, make up an emission spectrum. Each element's emission spectrum is unique.
The following two images are the emission spectrum of H

and Fe.
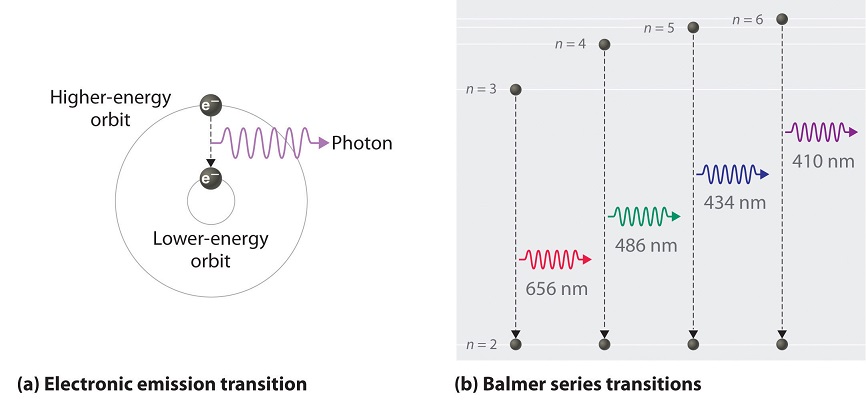
When the electrons in the atom are excited, for example by being heated, the additional energy pushes the electrons to higher energy orbitals. When the electrons fall back down and leave the excited state, energy is re-emitted in the form of a photon. The wavelength (or equivalently, frequency) of the photon is determined by the difference in energy between the two states. These emitted photons form the element's spectrum.
In the following experiment, electric current is passed through atoms to excite the electrons within:

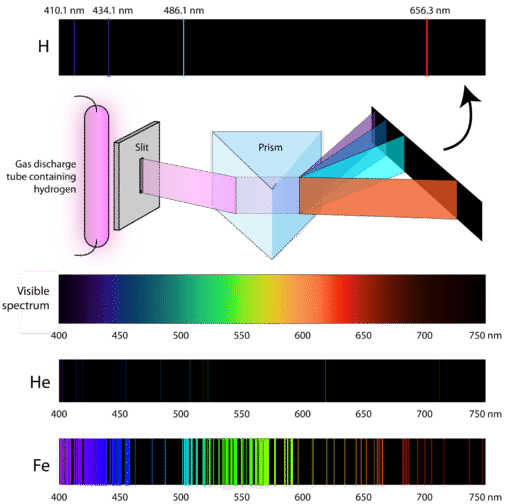
and since \[E_{\text{photon}}=hv\] where \[h\] is Planck's constant and \[v\] is the frequency, we conclude that only photons with specific energies are emitted by the atom. This proves that both energy levels exist, and the energy levels for each atom is different.