Le Chatelier's principle
Le Chatelier's principle
Le Chatelier's principle , also called Chatelier's principle (or the Equilibrium Law), is a principle of chemistry used to predict the effect of a change in conditions on chemical equilibrium.
Le Chatelier's principle can be stated as follows: A change in one of the variables that describe a system at equilibrium produces a shift in the position of the equilibrium that counteracts the effect of this change.
Changes in the concentrations of the reactants or products of a reaction shift the position of the equilibrium, but do not change the equilibrium constant for the reaction. Similarly, a change in the pressure on a gas-phase reaction shifts the position of the equilibrium without changing the magnitude of the equilibrium constant. Changes in the temperature of the system, however, affect the position of the equilibrium by changing the magnitude of the equilibrium constant for the reaction.
Misconceptions
A major misconception with the Le Chatelier's principle is that the systems "wants" something. Assume we have following reversible reaction where everything is in gas phase, \[\ce{A + B <=> C}\]. We'll simplify the rate law of the forward reaction to \[r_{f}=k_{f}[\ce{A}][\ce{B}]\] and the reverse rate law to \[r_{r}=k_{r}[\ce{C}]\].
Say, for instance if the volume is doubled while the amount of reactants stay the same, i.e. decreasing the concentration by half, the new forward rate will be quartered \[r_{f}^{\prime}=k_{f}\left( \frac{1}{2}[\ce{A}] \right)\left( \frac{1}{2}[\ce{B}] \right)\], while the new reverse rate will be halved \[r_{r}^{\prime}=k_{r}\left( \frac{1}{2}[\ce{C}] \right)\]. So, the reaction "shifts to the reactants", i.e. the concentration of \[\ce{A}\] and \[\ce{B}\] will increase while \[\ce{C}\] decreases.
As an extension, if volume is halved, the forward rate become quadrupled while the reverse rate is doubled, thus the reaction "shifts to products". Now, what happens when an inert gas is added? Since the inert gas appears in neither the forward nor reverse rate law, nothing is changed.
Changes in concentration
Changing the concentration of a chemical will shift the equilibrium to the side that would counter that change in concentration. The chemical system will attempt to partly oppose the change affected to the original state of equilibrium. In turn, the rate of reaction, extent, and yield of products will be altered corresponding to the impact on the system.
Consider the following system at a constant 500 C temperature: \[\ce{N2 + 3 H2 <=> 2 NH3}\]. If we increase the concentration of \[\ce{N2}\] by a factor of ten, stress will be placed onto the system. Thus, the system responds by minimizing the effect of this stress, by shifting the equilibrium toward the products (increased amounts of \[\ce{NH3}\] produced). Likewise, the subtraction of \[\ce{N2}\] would cause the reaction to "fill the gap" and shift the equilibrium to the reactants (decreased amounts of \[\ce{NH3}\] produced). This observation is supported by the Collision theory.
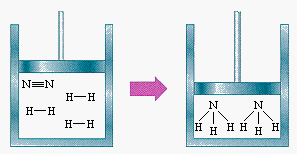
Another similar example:
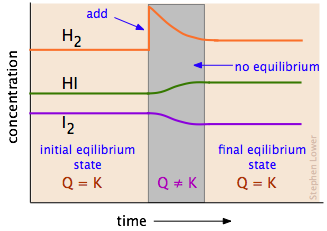
Changes in pressure
The equilibrium concentrations of the products and reactants do not directly depend on the total pressure of the system. They may depend on the partial pressure of the products and reactants, but if the number of moles of gaseous reactants is equal to the number of moles of gaseous products, pressure has no effect on equilibrium. Instead, the effect of changing the pressure on a gas-phase reaction depends on the stoichiometry of the reaction.
Using the same reaction example above, let's say we compress the system by a factor of 10. The reaction at equilibrium is now subjected to a stress, which is an increase in the total pressure on the system. The reaction now shifts in the direction that minimizes the effect of this stress. Thus, the reaction will shift towards the products (increase in concentration of \[\ce{NH3}\]) because this reduces the number of particles in the gas, thereby decreasing the total pressure on the system.
Essentially, an increase of pressure will favour the side of the reaction with lesser moles to decrease the number of particles in the system, therefore reducing pressure.
Changes in temperature
Chemical reactions either give off heat to their surroundings or absorb heat from their surroundings. If we consider heat to be one of the reactants or products of a reaction, we can understand the effect of changes in temperature on the equilibrium. Increasing the temperature of a reaction that gives off heat is the same as adding more of one of the products of the reaction. It places a stress on the reaction, which must be alleviated by converting some of the products back to reactants.
The reaction in which \[\ce{NO2}\] dimerizes to form \[\ce{N2O4}\] provides an example of the effect of changes in temperature on the equilibrium constant for a reaction. This reaction is exothermic.
\[\ce{2 NO2 <=> N2O4},\Delta H^{\circ}_{f}=-57.20\text{kJmol}^{-1}\], since this reaction is exothermic, therefore \[\ce{2 NO2 <=> N2O4 + \text{heat}}\].
If the temperature were increased, the heat content of the system would increase, so the system would consume some of that heat by shifting the equilibrium to the left, thereby producing more \[\ce{NO2}\] to decrease the temperature effect, as the reverse reaction, \[\ce{N2O4 -> 2 NO2}\] is an endothermic reaction (absorbs heat). More \[\ce{N2O4}\] would be produced if the reaction were run at a lower temperature.
The effect of temperature on equilibria, however, involves a change in the equilibrium constant as the dependence of \[K\] on temperature is determined by the sign of \[\Delta H\].
Effects of a catalyst
Catalysts only lower the activation energy and speeds up reaction, allowing the system to reach dynamic equilibrium faster, however it does not improve the yield of products.
Referenced by:
No backlinks found.