chemical equilibrium
Chemical equilibrium
Chemical equilibrium is the state in which both the reactants and products are present in concentrations which have no further tendency to change with time, so that there is no observable change in the properties of the system. This state results when the forward reaction proceeds at the same rate as the reversible reaction. The reaction rates of the forward and backward reactions are generally not zero, but they are equal. Thus, there are no net changes in the concentrations of the reactants and products. Such a state is also known as dynamic equilibrium (note that chemical equilibrium is a type of dynamic equilibrium).
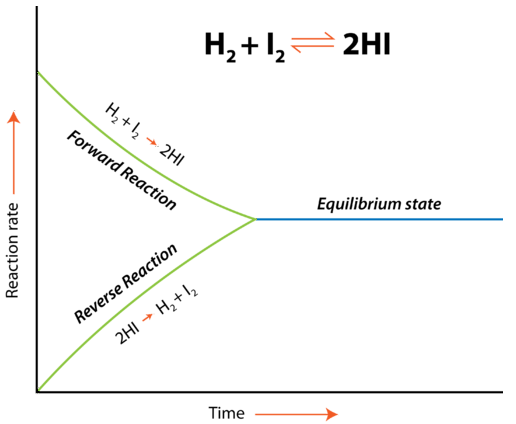
Taking \[\ce{H2 + I2 <=> 2 HI}\] as an example:
Initially, only the forward reaction occurs because no \[\ce{HI}\] is present. As soon as some \[\ce{HI}\] has formed, it begins to decompose back into \[\ce{H2}\] and \[\ce{I2}\]. Gradually, the rate of the forward reaction decreases while the rate of the reverse reaction increases. Eventually, the rate of combination of \[\ce{H2}\] and \[\ce{I2}\] to produce \[\ce{HI}\] becomes equal to the rate of decomposition of \[\ce{HI}\] into \[\ce{H2}\] and \[\ce{I2}\]. When the rates of the forward and reverse reactions have become equal to one another, the reaction has achieved a state of balance. Chemical equilibrium is the state of a system in which the rate of the forward reaction is equal to the rate of the reverse reaction.
In another example reaction of \[\ce{N2O4 <=> 2 NO2}\], the following graphs indicate how the composition of this system would vary as a function of time at a constant temperature. If the initial concentration of \[\ce{NO2}\] were zero, then it increases as the concentration of \[\ce{N2O4}\] decreases. Eventually the composition of the system stops changing with time, and chemical equilibrium is achieved. Conversely, if we start with a sample that contains no \[\ce{N2O4}\] but an initial \[\ce{NO2}\] concentration twice the initial concentration of \[\ce{N2O4}\], in accordance with the stoichiometry of the reaction, we reach exactly the same equilibrium composition. Thus equilibrium can be approached from either direction in a chemical reaction.
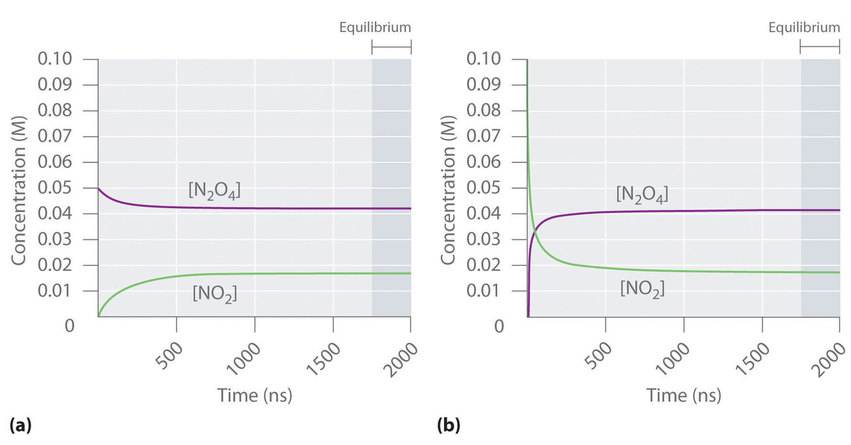
(a) Initially, this system contains 0.05 M of gaseous \[\ce{N2O4}\] and no gaseous \[\ce{NO2}\]. The concentration of \[\ce{N2O4}\] decreases with time as the concentration of \[\ce{NO2}\] increases. (b) Initially, this system contains 0.1 M \[\ce{NO2}\] and no \[\ce{N2O4}\]. The concentration of \[\ce{NO2}\] decreases with time as the concentration of \[\ce{N2O4}\] increases. In both cases, the final concentrations of the substances are the same: \[\ce{N2O4}\] = 0.0422 M and \[\ce{NO2}\] = 0.0156 M at equilibrium.