activation energy
Activation energy
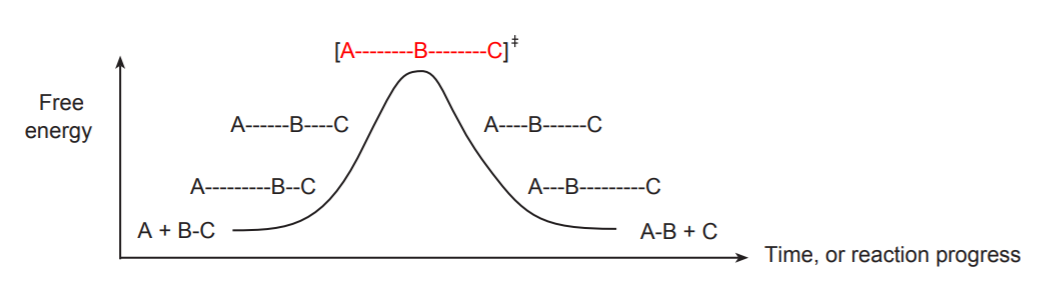
In the Arrhenius model of reaction rates, activation energy, \[E_{a}\] is the minimum amount of energy that must be available to reactants for a chemical reaction to occur.
When molecules collide, the kinetic energy of the molecules can be used to stretch, bend, and ultimately break bonds, leading to chemical reactions. If molecules move too slowly with little kinetic energy, or collide with improper orientation, they do not react and simply bounce off each other. However, if the molecules are moving fast enough with a proper collision orientation, such that the kinetic energy upon collision is greater than the minimum energy barrier, then a reaction occurs. The minimum energy requirement that must be met for a chemical reaction to occur is the activation energy.
Transition state
The transition state of is the state corresponding to the highest potential energy along the reaction coordinate. It represents a point of such high energy that it cannot possibly exist for any length of time.