equilibrium constant
Equilibrium constant
The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical equilibrium, a state approached by a dynamic chemical system after sufficient time has elapsed at which its composition has no measurable tendency towards further change.
For a system undergoing a reversible reaction, \[\ce{aA + bB +}\cdots \ce{<=>} \ce{cC + dD +}\cdots\], the thermodynamic equilibrium constant, would be \[K_{c}=\frac{[C]^{c}[D]^{d}\cdots}{[A]^{a}[B]^{b}\cdots}\], where the square brackets represent it's concentrations at equilibrium, with units \[\text{mol dm}^{-3}\].
For partial pressures, the equilibrium constant, would be \[K_{p}=\frac { (P_{C})^{c} (P_{D})^{d }} { (P_{A})^{a} (P_{B})^{b} }\]. For the calculation of partial pressures, we usually convert it into mole fraction first.
Do note that square brackets are only used for writing \[K_{c}=\frac{[\ce{NO2}]^{2}}{[\ce{NO}]^{2}[\ce{O2}]}\]. After substituting the concentrations, we use curved brackets.
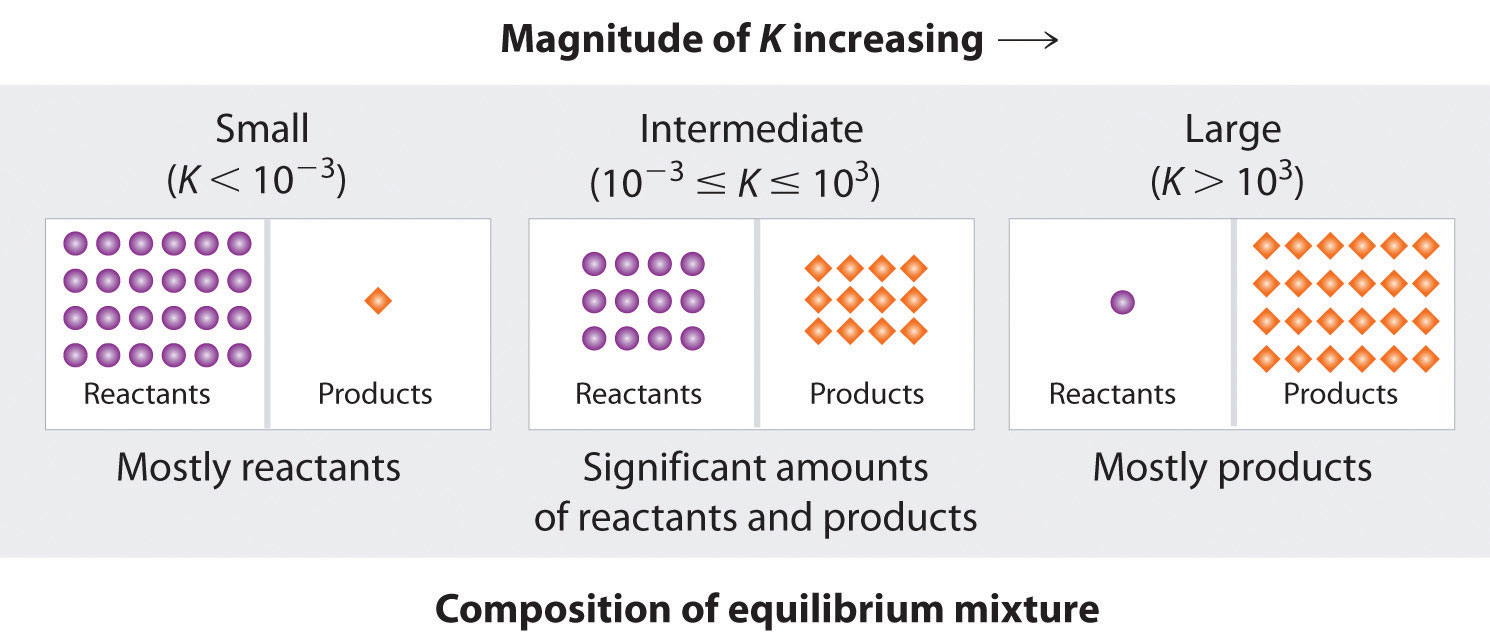
The equilibrium constant can vary over a wide range of values.
- When \[K_{c}\] is close to 1, the concentrations of reactants and products are similar, the equilibrium lies near the centre.
- When \[K_{c}>1\], the reaction will favour the products and the equilibrium position lies to the right, whereas when \[K_{c}\] goes beyond \[10^{5}\], we can assume the reaction is near completion.
- When \[K_{c}<1\], the reaction will favour the reactants and the equilibrium position lies to the left, whereas when \[K_{c}\] is lesser than \[10^{-5}\], we can assume the reaction hardly occurs at all.
Calculating equilibrium constants without complete information
Example 1
Let's assume we have a 2dm3 solution containing 1 mol of \[\ce{NOCl}\] placed in a reactor and heated to 227 Celsius until the system reaches equilibrium. The contents of the reactor were then analyzed and was found to contain 0.056 mol of \[\ce{Cl2}\]. The equation for the decomposition is \[\ce{2NOCl(g) <=> 2NO(g) + Cl2(g)}\]. Calculate the equilibrium constant at this temperature.
First, we establish that the equilibrium constant, \[K_{c}\], equals \[\frac{[\ce{NO}]^{2}[\ce{Cl2}]}{[\ce{NOCl}]^{2}}\], and the initial molar concetration of \[\ce{NOCl}\] is \[\frac{1\text{mol}}{2\text{dm}^{3}}=0.5\text{moldm}^{-3}\]. We then construct a table:
| \[\ce{NOCl}\] | \[\ce{NO}\] | \[\ce{Cl2}\] | |
| Initial | 0.5 | 0 | 0 |
| Change | -0.056 | +0.056 | +0.028 |
| Final | 0.444 | 0.056 | 0.028 |
To break it down:
- Since the system contains 0.056 mol of \[\ce{Cl2}\], then the molar concentration would be 0.028 moldm-3, as the system has a volume of 2dm3
- Now that we know the molar concentration of \[\ce{Cl2}\] at equilibrium, according to the reaction, the concentration of \[\ce{NO}\] must be twice of \[\ce{Cl2}\], thus being 0.056 moldm-3
- Same goes with the molar concentration of \[\ce{NOCl}\], however, since some of the \[\ce{NOCl}\] molecules have reacted to form \[\ce{NO}\] and \[\ce{Cl2}\], thus it's concentration must decrease
Thus, \[K=\frac{[\ce{NO}]^{2}[\ce{Cl2}]}{[\ce{NOCl}]^{2}}=\frac{0.056^{2}\cdot0.028}{0.444^{2}}=4.5\times 10^{-4}\].
Example 2
The water-gas shift reaction is important in several chemical processes, such as the production of \[\ce{H2}\] for fuel cells. This reaction can be written as follows: \[\ce{H2(g) + CO2(g) <=> H2O(g) + CO(g)}\] and \[K=0.106\] at 700 Kelvin. If a mixture of gases that initially contains 0.0150 moldm-3 \[\ce{H2}\] and 0.0150 moldm-3 \[\ce{CO2}\] is allowed to equilibrate at 700 Kelvin, What are the final concentrations of all substances present?
Same as the previous example, we first establish that the equilibrium constant, \[K=\frac{[\ce{H2O}][\ce{CO}]}{[\ce{H2}][\ce{CO2}]}\], and we let \[x\] be the change in concentration of \[\ce{H2O}\].
| \[\ce{H2}\] | \[\ce{CO2}\] | \[\ce{H2O}\] | \[\ce{CO}\] | |
| Initial | 0.0150 | 0.0150 | 0 | 0 |
| Change | -\[x\] | -\[x\] | +\[x\] | +\[x\] |
| Final | \[0.0150-x\] | \[0.0150-x\] | \[x\] | \[x\] |
Since in the reaction all of the reactants and should have all the same concentrations, all the changes to the molar concentrations should be increased or decreased by \[x\].
Thus, \[K=\frac{x\cdot x}{(0.015-x)\cdot(0.015-x)}=0.106\]:
Hence, with our \[x\] value we can find that \[[\ce{H2}]\] and \[[\ce{CO2}]\] equals to \[0.011316\,\text{moldm$^{-3}$}\] and \[[\ce{H2O}]\] and \[[\ce{CO}]\] equals to \[0.003684\,\text{moldm$^{-3}$}\].
Example 3
In an experiment carried out, hydrogen iodide was found to be 22.3% dissociated at 730.8 Kelvin. Calculate \[K_{c}\] for \[\ce{2HI(g) <=> H2(g) + I2}\].
The way to solve this is quite straightforward. Since no explicit molar concentrations are given, but we do know that for every \[0.223n\] moles of each product formed, \[1-0.223n=0.777n\] moles of \[\ce{HI}\] remains. Then,
Example 4
Ordinary white phosphorus, \[\ce{P4}\], forms a vapour which dissociates into diatomic molecules at high temperatures: \[\ce{P4(g) <=> 2P2(g)}\]. A sample of white phosphorus, when heated at 1000 Celsius (1273 Kelvin), formed a vapour having a total pressure of 0.20 atm and a density of 0.152 grams per litre at equilibrium. Note that 1 atm = 101325 Pa. Find the equilibrium constant.
In this scenario, we first calculate the mole fractions of each reactant, then find its respective pressures.
| \[\ce{P4(g)}\] | \[\ce{P2(g)}\] | |
| Initial | 1 | 0 |
| Change | \[-x\] | \[2x\] |
| Equilibrium | \[1-x\] | \[2x\] |
| Mole fraction | \[\chi\ce{P4}=\frac{1-x}{1-x+2x}\] | \[\chi\ce{P2}=\frac{2x}{1-x+2x}\] |
| Equilibrium (pressures) | \[p\ce{P4}=\chi\ce{P4}\cdot0.2\] | \[p\ce{P2}=\chi\ce{P2}\cdot0.2\] |
What the bottom row is doing is essentially just multiplying the mole fraction of each gas by the total pressure, i.e. \[p_{i}=\chi_{i}P_{\text{tot}}\].
Expressing the equilibrium constant in terms of \[x\] now is \[K_{p}=\frac{(P_{\ce{P2}})^{2}}{(P_{\ce{P4}})}=\frac{\left( \frac{2x}{1+x} \right)^{2}\cdot0.2^{2}}{\left( \frac{1-x}{1+x} \right)\cdot0.2}=\left( \frac{4x^{2}}{1-x^{2}} \right)0.2\]. We're not done yet however.
Example 5
Phosgene (\[\ce{COCl2}\]) is a poisonous gas that dissociates at high temperature into two other poisonous gases, carbon monoxide and chlorine. The equilibrium constant \[K_{p}=0.0041\] at 600 Kelvin. Find the equilibrium composition of the system after 0.124 atm of phosgene is allowed to reach equilibrium at this temperature.
The chemical reaction that happens is \[\ce{COCl2 <=> CO(g) + Cl2(g)}\].
| \[\ce{COCl2}\] | \[\ce{CO}\] | \[\ce{Cl2}\] | |
| Initial (pressure) | 0.124 atm | 0 | 0 |
| Change (pressure) | \[-x\] | \[+x\] | \[+x\] |
| Equilibrium (pressure) | \[0.124-x\] | \[+x\] | \[+x\] |
Substitution of the equilibrium expression gives us \[\frac{x^{2}}{0.124-x}=0.0041\implies x\approx0.0206\].