rate of reaction
Rate of reaction
The reaction rate or rate of reaction is the speed at which a chemical reaction takes place, defined as proportional to the increase in the concentration of a product per unit time and to the decrease in the concentration of a reactant per unit time.
\[\text{rate}=\frac{\Delta \text{concentration}}{\text{time}}\], and for a reaction of the form \[\ce{A + 3 B -> 2 C}\], the rate can be expressed as \[\text{rate}=-\frac{\Delta [A]}{1\Delta t}=-\frac{\Delta [B]}{3\Delta t}=\frac{\Delta [C]}{2\Delta t}\] (this form is also known as the rate expression). The concentration of a reactant always decreases with time, so \[\Delta[A]\] and \[\Delta[B]\] are both negative. Since negative rates do not make much sense, rates expressed in terms of a reactant concentration are always preceded by a minus sign to make the rate come out positive. It is also clear that \[[B]\] decreases three times as rapidly as \[[A]\], so in order to avoid ambiguity when expressing the rate in terms of different components, it is customary to divide each change in concentration by the appropriate coefficient.
Additionally, if we multiply the stoichiometric coefficient, e.g. \[\ce{2A + 6B -> 4C}\], the rate of reaction would be \[\text{rate}=-\frac{\Delta [A]}{2\Delta t}=-\frac{\Delta [B]}{6\Delta t}=\frac{\Delta [C]}{4\Delta t}\]. This expression and our originally expression are both numerically equivalent, i.e. when you plug in the actual concentration changes into both equations, you will get the exact same rate of reaction. Thus, we can say that multiplying the coefficients of the reaction will not change the rate expression.
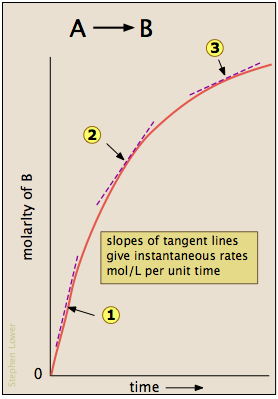
Mean, initial rate, and instantaneous rate
- Mean: Calculated by taking the \[\frac{\text{Change in product or reactant}}{\text{time}}\].
- Initial rate: Calculated by drawing a line tangent to the point \[t=0\] and calculating the gradient.
- Instantaneous rate: Calculated by drawing a line tangent to the point \[t\] and calculating the gradient.