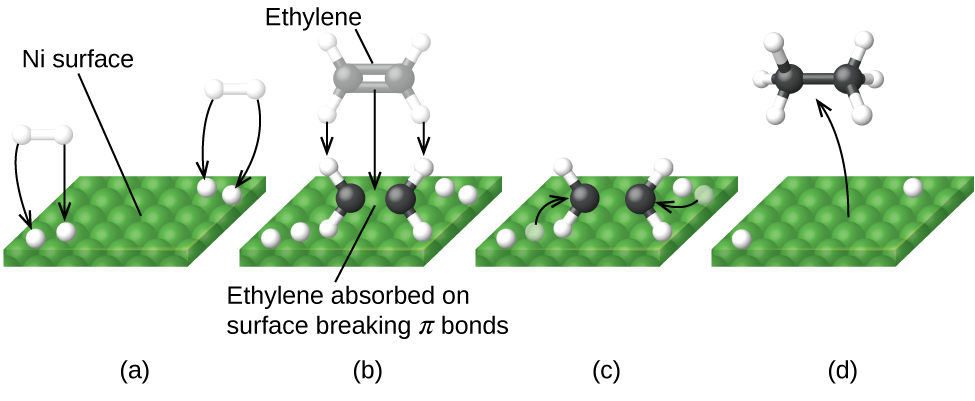
Collision theory
Collision theory
Collision theory is a principle of chemistry used to predict the rates of chemical reactions. It states that when suitable particles of the reactant hit each other with the correct orientation, only a certain amount of collisions result in a perceptible or notable change; these successful changes are called successful collisions. The successful collisions must have enough energy, also known as activation energy, at the moment of impact to break the pre-existing bonds and form all new bonds. This results in the products of the reaction.
Postulates
- The rate of reaction is proportional to the rate of reactant collisions: \[\text{reaction rate}\propto \frac{\text{number of collision}}{\text{time}}\], or \[\text{reaction rate}\propto \frac{\text{change in concentration}}{\text{time}}\].
-
The reacting species must collide in an orientation that allows contact between the atoms that will become bonded together in the product.
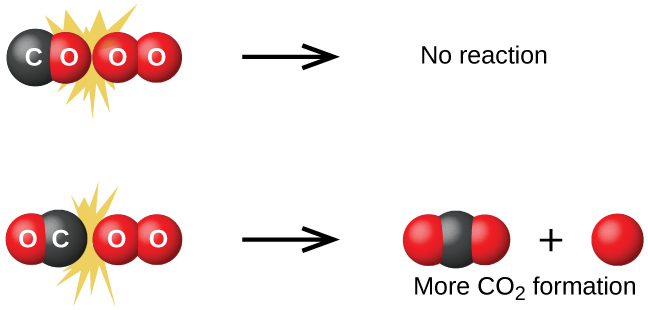
- The collision must occur with adequate energy to permit mutual penetration of the reacting species' valence shells so that the electrons can rearrange and form new bonds.
Factors affecting the rate of reaction
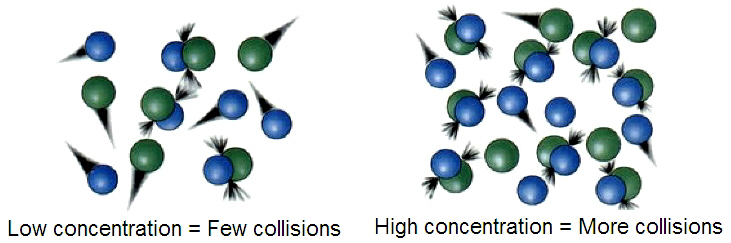
- A high concentration or pressure means a large number of particles in a small volume. This increases the chance of particles colliding with the correct orientation, increasing the rate of reactant collision thus increasing the reaction rate.
- At a higher temperature, particles have more kinetic energy. More particles are likely to have energy higher than the activation energy, thus more effective at breaking bonds. In an environment of high temperature, particles will also move around faster, making them collide more frequently.
-
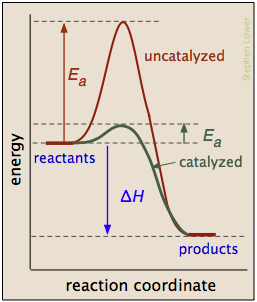
The presence of a catalyst increases the rate of reaction by providing a reaction pathway with lower activation energy.