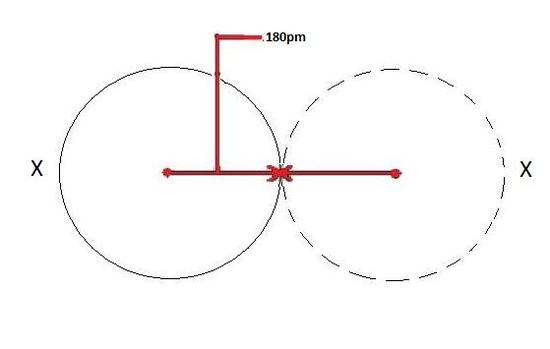
metallic radius
Metallic radius
The metallic radius is defined as one-half of the distance between the two adjacent metal ions in the metallic structure. The radii follow general periodic trends: they decrease across the period due to the increase in the effective nuclear charge, which is not offset by the increased number of valence electrons; but the radii increase down the group due to an increase in the principle quantum number.