wavefunction
Wavefunctions
- A wavefunction, \[\psi\], is a mathematical function that relates the location of an electron at a given point in space (identified by x, y, and z coordinates) to the amplitude of its wave, which corresponds to its energy. Thus each wavefunction is associated with a particular energy \[E\].
- A wavefunction uses three variables to describe the position of an electron. A fourth variable is usually required to fully describe the location of objects in motion. Three specify the position in space (as with the Cartesian coordinates x, y, and z.), or in quantum mechanics, the three quantum numbers, Principle quantum number, Azimuthal quantum number and Magnetic quantum number, and one specifies the time at which the object is at the specified location.
-
The square of the wavefunction, \[\psi^{2}\] at a given point is proportional to the probability of finding an electron at that point, which leads to a distribution of probabilities in space. (which is also what atomic orbitals are based on)
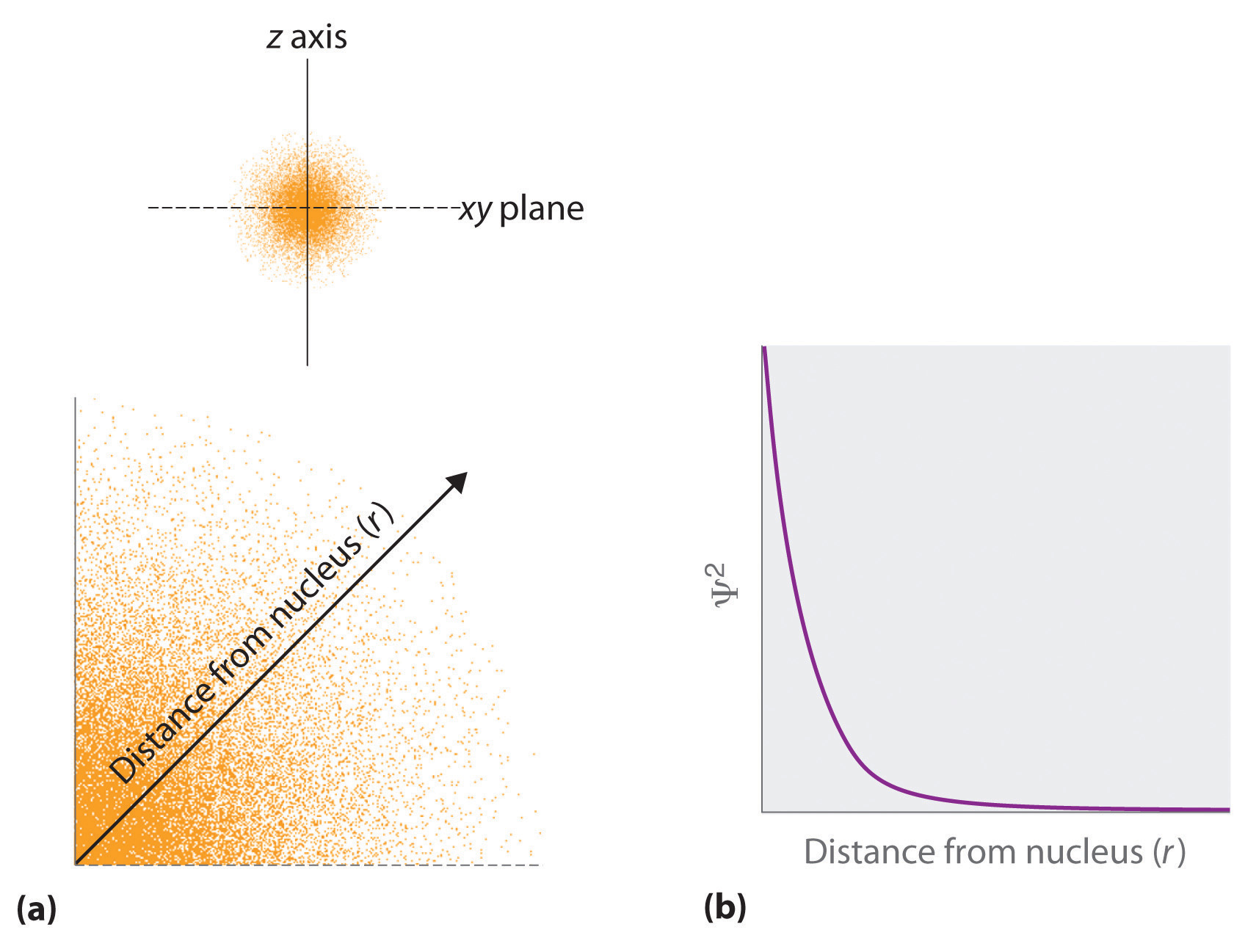
(a) The density of the dots shows electron probability. (b) In this plot of ψ2 versus r for the ground state of the hydrogen atom, the electron probability density is greatest at r=0 (the nucleus) and falls off with increasing r. Because the line never actually reaches the horizontal axis, the probability of finding the electron at very large values of r is very small but not zero.
Examples
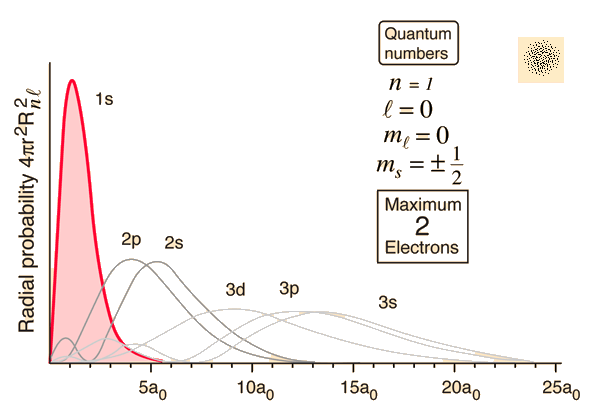
The normalized hydrogen wavefunctions of different orbitals in a hydrogen atom (Source):