kinetic theory of gases
Kinetic theory of gases
The kinetic theory of gases is a simple classical model of the thermodynamic behavior of gases. It treats a gas as composed of numerous particles, too small to be seen with a microscope, in constant, random motion.
Evidence for the kinetic theory
A crucial observation is that although solids and liquids are nearly incompressible, gases can be compressed. This makes us hypothesize that the atoms in a solid are packed tightly, while a gas is mostly vacuum with large spaces between molecules. Liquids and solids usually have densities about a thousand times greater than most gases, so evidently each molecule in a gas is separated from its nearest neighbours by a space about ten times the size of the molecules themselves.
Now, this begs the question that, if gas molecules have nothing but empty space between them, why don't the molecules in the room just fall to the floor? The only possible answer is that they are in rapid motion, continually rebounding from the walls, floor and ceiling. When this theory was proposed by Daniel Bernoulli, it was met with considerable opposition because it seemed as though the molecules in a gas would eventually calm down and settle into a thin film on the floor. There was no precedent for this kind of perpetual motion. No rubber ball, however elastic, rebounds from a wall with exactly as much energy as it originally had, nor do we ever observe a collision between balls in which none of the kinetic energy at all is converted to heat and sound. The analogy is a false one, however. A rubber ball consists of atoms, and when it is heated in a collision, the heat is a form of motion of those atoms. An individual molecule, however, cannot possess heat. Likewise sound is a form of bulk motion of molecules, so colliding molecules in a gas cannot convert their kinetic energy to sound. Molecules can indeed induce vibrations such as sound waves when they strike the walls of a container, but the vibrations of the walls are just as likely to impart energy to a gas molecule as to take energy from it. Indeed, this kind of exchange of energy is the mechanism by which the temperatures of the gas and its container become equilibrated.
Assumptions
The five basic tenets of the kinetic-molecular theory are as follows:
- A gas is composed of molecules that are separated by average distances that are much greater than the sizes of the molecules themselves. The volume occupied by the molecules of the gas is negligible compared to the volume of the gas itself.
- The molecules of an ideal gas exert no attractive forces on each other, or on the walls of the container.
- The molecules are in constant random motion, and as material bodies, they obey Newton's laws of motion, i.e. the molecules move in straight lines until they collide with each other or with the walls of the container.
- Collisions are perfectly elastic, i.e. when two molecules collide, they change their directions and kinetic energies, but the total kinetic energy is conserved. There are also no three-body or higher interactions.
- The average kinetic energy of the gas molecules is directly proportional to the absolute temperature. It is important to note of the term "average" here, as the velocities and kinetic energies of individual molecules will span a wide range of values, and some will even have zero velocity at a given instant.
Molecular velocities and kinetic energy
In a gas sample, individual molecules have widely varying speeds; however because of the vast number of molecules and collisions involved, the molecular speed distribution and average speed are constant. This distribution is known as a Maxwell-Boltzmann distribution, and it depicts the relative numbers of molecules in a bulk sample of gas that possesses a given speed.
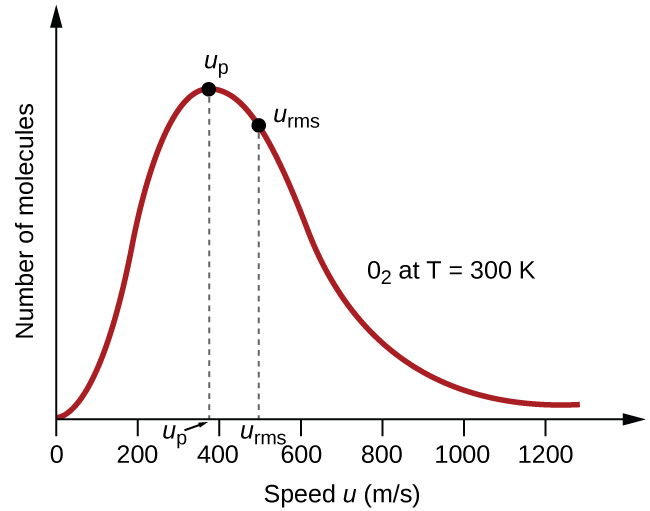
This graph shows the molecular speed distribution for oxygen gas at 300 Kelvins. The most probable speed of a molecule is denoted by \[u_{p}\], which is slightly lesser than 400 meters per second. The average molecular speed, \[u_{\text{rms}}\], also known as the root mean square speed is instead closer to 500 meters per second.
The kinetic energy of a particle going at speed \[u\] is given by \[\frac{1}{2}mu^{2}\]. However, since we're dealing with a large number of gas molecules, it's quite unrealistic to calculate the kinetic energy of every single gas molecules. So, we introduce an average to the speed, i.e. \[u_{\text{rms}}\]. It's defined as the square root of the average of the squares of the velocities: \[u_{\text{rms}}=\sqrt{\overline{u^{2}}}=\sqrt{\frac{u_{1}^{2}+u_{2}^{2}+u_{3}^{2}+\cdots}{n}}\], where \[\overline{u^{2}}\] is the mean of the square of velocities and \[n\] equals to the number of particles. With this defined, the average kinetic energy is then equal to \[\frac{1}{2}mu^{2}_{\text{rms}}\]. Note that \[\left\langle x \right\rangle\] and \[\overline{x}\] are generic average/mean symbols, i.e. \[x_{\text{rms}}=\sqrt{\left\langle x^{2} \right\rangle}=\overline{x^{2}}\], also make sure not to mix up \[\overline{x}^{2}\ne\overline{x^{2}}\] and \[\sqrt{\overline{x^{2}}}\ne \overline{x}\].
For this equation to have any use in any defining temperature, gas molecules must have a completely random motion, a condition known as thermal equilibrium. At thermal equilibrium, the system has no "memory" of how it was prepared and the only forces acting on the particles are the contact forces felt during collisions with the walls of the gas container and with other molecules. Then, by equipartition theorem, the temperature may be defined as a quantity directly proportional to the average kinetic energy of the gas molecules, \[E_{\text{k,avg}}=\frac{1}{2}mu^{2}_{\text{rms}}=\frac{3}{2}k_{\text{B}}T\], where \[k_{\text{B}}\] is the Boltzmann constant.
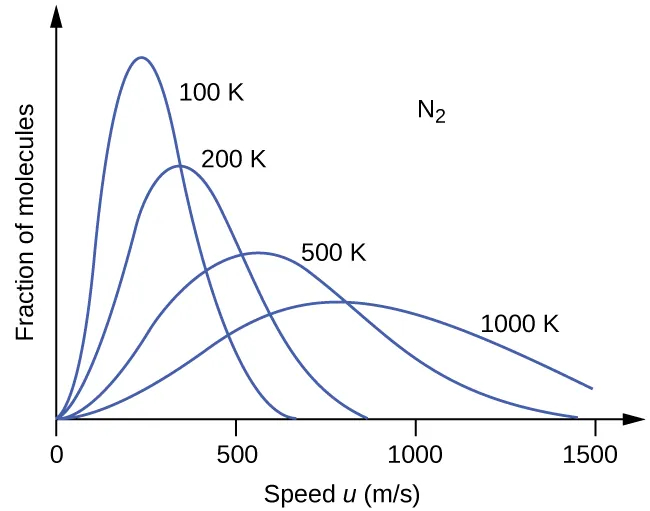
With this relationship established, we can predict that if the temperature of a gas increases, its \[\text{KE}_{\text{avg}}\] increases, thus more molecules have higher speeds and fewer molecules have lower speeds.
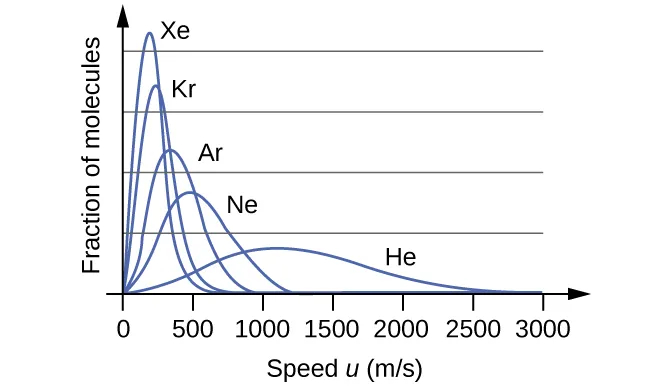
Another important observation is that since regardless of mass, \[E_{\text{k,avg}}=\frac{3}{2}k_{\text{B}}T\], at a given temperature, all gases will have the same \[\text{KE}_{\text{avg}}\] for their molecules. Gases composed of lighter molecules will have more high-speed particles, with a speed distribution that peaks at relatively higher velocities. Similarly, heavier molecules will have more low-speed particles, with a speed distribution that peaks at low velocities.