Azimuthal quantum number
Azimuthal quantum number
In quantum mechanics, the azimuthal quantum number \[\ell\] is a quantum number for an atomic orbital that determines its orbital angular momentum and describes aspects of the angular shape of the orbital. Each different angular momentum states can take \[2(2\ell+1)\] electrons. This is because each distinct orbital can be occupied by 2 electrons, and the Magnetic quantum number, \[m_{\ell}\] runs from \[-\ell\] to \[\ell\] in integer units, giving a total of \[2\ell+1\] states.
Quantum subshells for the azimuthal quantum number:

Spherical harmonics:
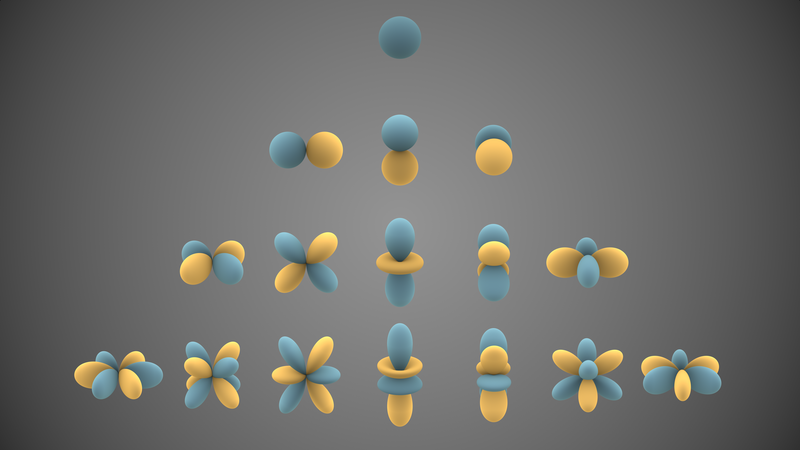
Orbital angular momentum
In quantum mechanics, the classical concept of trajectory of a particle is abandoned. The quantum state of a particle e.g. the electron is characterized by a wavefunction which is nothing but a function in space and time \[\psi(x,y,z,t)\] that contains all information about the particle and that extends over space. The simplest case where angular momentum appears in the context of quantum mechanics is the one of a particle in a “ring”. Now you have a wave function which extends over a circular region of space which implies that your particle has angular momentum. When you apply the same idea to the Hydrogen atom you will find the orbital angular momentum.
Then the entire idea of "electron moving around the atom" needs to be abandoned if we want to describe the quantum-mechanical notion of it. However, there is still a mapping of classical angular momentum onto a quantum one, and it retains the idea of angular dependence. So the quantum angular momentum describes how the probability distribution to find the electron at certain positions (that is what replaces in quantum mechanics the classical position of the electron), is "skewed angularly" about some center. In atomic context, the \[s\] orbitals have zero angular momentum, which means that they don't have any angular dependency, and the probability to find the electron when it is in this shell is uniform in any direction. The states with nonzero angular momentum will have a probability distribution that prefers some directions to others.
One feature that can be taken directly from classical angular momentum to angular one, is that only electrons with no angular momentum (that is \[\ell=0\], or \[s\] orbitals), have any probability to be found right in the center. For any orbital with \[\ell>0\] the electron has zero probability to be found at the center of the atom, which indeed sits well with out classical intuition of something that revolves around the center.