Spin Magnetic quantum number
Spin magnetic quantum number
Spin is a quantum property that has no real analogy that would make sense to us on a macroscopic level. The spin of an electron is an intrinsic property, meaning it does not arise from any physical motion. However, we do know that spin has associated with it magnetic properties.
In physics, the spin quantum number is a quantum number (designated \[s\]) that describes the intrinsic angular momentum (or spin angular momentum, or simply spin) of an electron or other particle. It has the same value for all particles of the same type, such as \[s=\frac{1}{2}\] for all electrons. It is an integer for all bosons, such as photons, and a half-odd-integer for all fermions, such as electrons and protons.
The component of the spin along a specified axis is given by the spin magnetic quantum number, conventionally written \[m_{s}\] The value of \[m_{s}\] is the component of spin angular momentum, in units of the reduced Planck constant \[\hbar\], parallel to a given direction (conventionally labelled the z-axis). It can take values ranging from \[+s\] to \[-s\] in integer increments. For an electron, \[m_{s}\] can be either \[+\frac{1}{2}\], represented by \[\big\uparrow\], or \[-\frac{1}{2}\], represented by \[\big\downarrow\].
Total Spin quantum number
The total spin quantum number can be obtained via \[\pm\frac{1}{2}\times\text{Number of unpaired electrons}\]. As according to Hund's rules all unpaired electrons will have parallel (same orientation) spins. Or more accurately, it is the sum of \[m_{s}\] values for each unpaired electron.
Spin multiplicity
The multiplicity is often equal to the number of possible orientations of the total spin relative to the total orbital angular momentum \[L\].
Spin multiplicity is calculated via \[2S+1\] where \[S\] is the total spin. States with multiplicity 1, 2, 3, 4, 5 are respectively called singlets, doublets, triplets, quartets and quintets.
Ex. Carbon ([He] 2s22p2) has two unpaired electrons, therefore it's spin multiplicity is \[2(2\cdot\frac{1}{2})+1=3\]. (Triplet state) which corresponds to the \[m_{s}\] values of -1,0,1. The ground state of carbon can also be written as \[^{3}P\].
Spin of electrons in magnetic fields (or not)
In the absence of a magnetic field (or other spin to couple to), individual electrons have random spins. It is purely conventional that we list electrons in atomic orbitals as \[+\frac{1}{2}\] first. However, that does not imply that unpaired electrons in an atom can have different spins as Hund's rules declares that the electrons in the orbital are filled up first by electrons whose spins are parallel to each other to minimize repulsion. Which means that it will either be filled with all up-spins or all down-spins first then opposite spins. Also, the final filled orbital state (all up or all down) is not completely random.
The two possible spin states are the same energetically unless a magnetic field is present. Once that situation arises, there is an energetic separation between the two states.
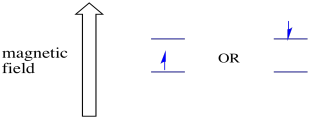
In the presence of a magnetic field, the two spin states separate into two different energy levels. The amount of separation between the energy levels depends on the magnitude of the magnetic field. The stronger the magnetic field, the greater the separation.
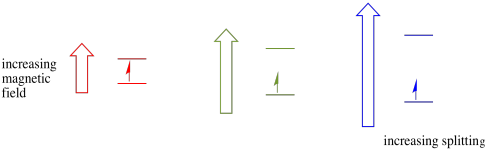
Reasons for different spins in electrons
The term spin is used to describe a property that electrons seem to have because their energy can have two different values when in a magnetic field. The only thing that we can imagine from a macroscopic point of view with this property is spin so that is what it was and is still called.
Electrons are fermions, and no two fermions can have the same state. (See elementary particle) A way of looking at this is to know that every pair of electrons have to be different somehow, and once you've got it down to a single orbital there is no other way for them to differ, so the spins must differ.