state function
State function
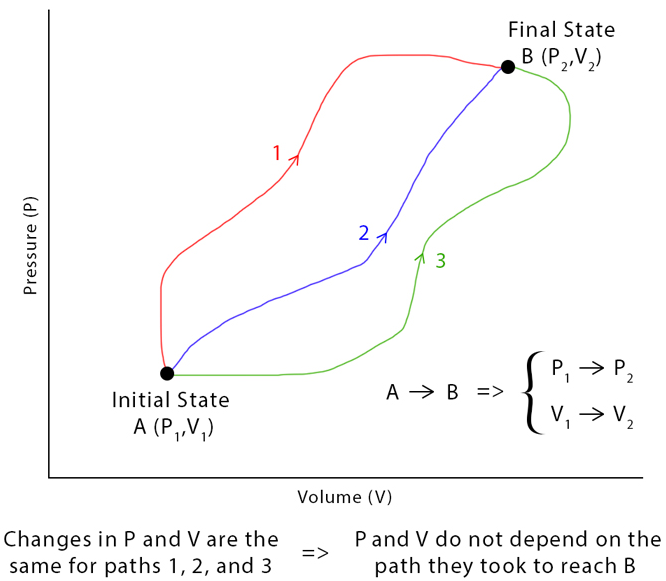
In thermodynamics, a state function for a thermodynamic system is a mathematical function relating several state variables/quantities (pressure, volume, temperature, mass, etc.) that depend only on the current equilibrium thermodynamic state of the system, not the path which the system has taken to reach that state.
In simpler words, a state function (or a function of state) is independent on the path by which one arrives to this state. It depends only on the state itself (more precisely on the state variables). For instance, if the state of a thermodynamic system is fully described by pressure and volume, any function that depends only on pressure and volume (such as the internal energy of an ideal gas) is a state function. On the other hand, heat or work are not state functions, since we can arrive to the same state by different paths, i.e. via transferring different amounts of heat and work.
Another way to think of state functions is as integrals. Integrals depend on only three things: the function, lower limit and upper limit, i.e. \[\int_{t_{0}}^{t_{1}}H(t)\,dt=H(t_{1})-H(t_{0})\], which is equivalent to a familiar definition of enthalpy. This illustrates how state functions depend only on the final and initial value and not on the object's history or the path taken to get from the initial to the final value.
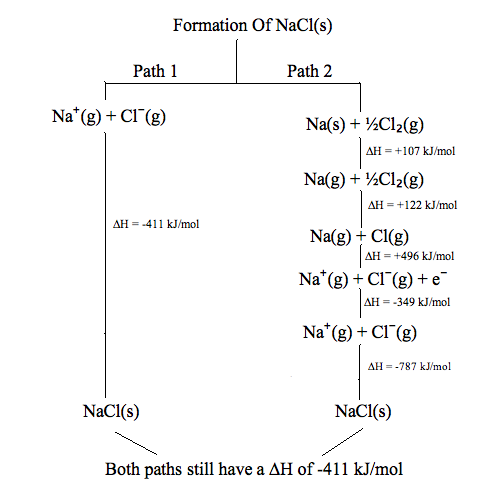
The figure below is a perfect example of a state function, no matter which path is taken to form \[\ce{NaCl}\], it results the same enthalpy of formation of \[-411\text{kJ mol$^{-1}$}\]. The key to this idea is that the change in enthalpy has nothing to do with the particulars of the process that brought the system from its initial state to the final state. The difference in enthalpy will always be the same.