organic compounds (naming)
Organic compounds (naming)
Prefixes
| Number of Carbons | Prefix |
|---|---|
| 1 | Meth |
| 2 | Eth |
| 3 | Prop |
| 4 | But |
| 5 | Pent |
| 6 | Hex |
| 7 | Hept |
| 8 | Oct |
| 9 | Non |
| 10 | Dec |
Naming rules
- Choose the longest, most substituted carbon chain containing a functional group.
- A carbon bonded to a functional group must have the lowest possible carbon number. If there are no functional groups, then any substitute present must have the lowest possible number.
- Take the alphabetical order into consideration; that is, after applying the first two rules given above, make sure that your substitutes and/or functional groups are written in alphabetical order.
Example:
- The longest chain here would be the seven carbon chain
- Carbons bonded to a functional group must have the lowest possible carbon number. After taking functional groups into consideration, any substitutes present must have the lowest possible carbon number. If we number the chain from the left, bromine, the methyl group and chlorine would occupy the second, fifth and sixth positions, respectively. If we number the chain from the right, chlorine, the methyl group and bromine would occupy the second, third and sixth positions. The position of the methyl, therefore, becomes a point of difference. To satisfy the second rule, we want to choose the numbering scheme that provides the lowest possible numbering of this substitute. Therefore, we will start numbering the chain from the right.
- After applying the first two rules, take the alphabetical order into consideration. Alphabetically, bromine comes before chlorine.
Thus, the name of this molecule would be 6-bromo-2-chloro-3-methylheptane.
Another few examples:
1,1,2-tribromopropane
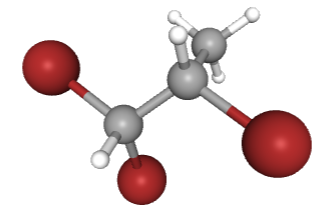
1,1,1-tribromo-2,2,2-trifluoroethane