Hess's law
Hess's law
Hess' law states that the change of enthalpy in a chemical reaction is the same regardless of whether the reaction takes place in one step or several steps, provided the initial and final states of the reactants and products are the same. In other words, if a chemical change takes place by several different routes, the overall enthalpy change is the same, regardless of the route by which the chemical change occurs (provided the initial and final condition are the same). If this were not true, then one could violate the first law of thermodynamics.
Example
Say we are given the equations:
By adding equation 1, 2 and 3 together, we get \[\ce{2 Al(s) + Fe2O3(s) -> Al2O3(s) + 2 Fe(s)},\Delta H=-851.1\text{kJ}\].
Additionally, if a reaction must be reversed, invert the sign of \[\Delta H\] for that reaction; if a reaction must be multiplied by a factor to obtain the correct number of moles of a substance, multiply its \[\Delta H\] value by that same factor.
Energy cycle diagram
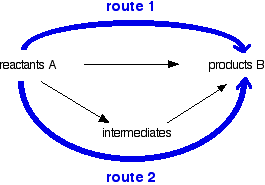
Hess's Law implies that the overall enthalpy change in these two routes will be the same.
It doesn't matter if we're given the enthalpy of combustion and told to find the enthalpy of formation, or just any combination of enthalpies for that matter. Since the enthalpies just describe energy, it does not differentiate between whatever enthalpy it is. As long as the equations are balanced, the change in energy must be the same.
Example 1
Now, assuming we want to know the enthalpy change of formation, \[\Delta H_{f}^{\circ}\], of benzene directly from its elements, carbon and hydrogen, i.e. \[\ce{6C (s) + 3H2 (g) -> C6H6 (l)}\]. This is regardless of how the benzene was synthesized, as the total change in energy should theoretically be the same.
We know that the enthalpy change of combustion, \[\Delta H_{c}^{\circ}\] for benzene is -3267 kJ per mol, whose reaction is \[\ce{C6H6 (l) + 7.5O2 (g) -> 6CO2 (g) + 3 H2O (l)}\]; \[\Delta H_{c}^{\circ}\] of carbon is -394 kJ per mol, whose reaction is \[\ce{C (s) + O2 (g) -> CO2 (g)}\]; \[\Delta H_{c}^{\circ}\] for hydrogen gas is -286 kJ per mol, whose reaction is \[\ce{H2 (g) + 0.5O2 (g) -> H2O (l)}\].
Drawing the energy diagram becomes as simple as connecting everything together:
The term \[6(-394)+3(-286)\] is derived from adding the enthalpy of combustion of carbon and hydrogen gas.
The reason why oxygen isn't included in here is that the oxygen gas consumed from the "combusting \[\ce{C}\] and \[\ce{H2}\]" route is exactly the same as the "forming benzene then combusting it" route. Thus, one can omit the oxygen gas for a simpler diagram as it has no effect on the calculation.
Finally, determining the enthalpy of formation is as simple as doing \[\Delta H_{f}^{\circ}=6(-394)+3(-286)-(-3267)=+45\,\text{kJ mol$^{-1}$}\].
Example 2
Assume we want to calculate the enthalpy change for the reaction, \[\Delta H_{c}^{\circ}\], between ethene and hydrogen chloride gases to make chloroethane gas, \[\ce{C2H4(g) + HCl(g) -> C2H5Cl(g)}\].
We are given that the enthalpy of formation, \[\Delta H_{f}^{\circ}\] of \[\ce{C2H4(g)}\] is +52.2 kJ per mol, whose reaction is \[\ce{2C(s) + 2H2(g) -> C2H4(g)}\]; \[\Delta H_{f}^{\circ}\] of \[\ce{HCl(g)}\] is -92.3 kJ per mol, whose reaction is \[\ce{0.5H2(g) + 0.5Cl2(g) -> HCl(g)}\]; \[\Delta H^{\circ}_{f}\] of \[\ce{C2H5Cl(g)}\] is -109 kJ per mol, whose reaction is \[\ce{2C(s) + 2.5H2(g) + 0.5Cl2(g) -> C2H5Cl(g)}\].
Again, to make these equations more intuitive, we draw an energy cycle:
Then, \[\Delta H_{r}^{\circ}=-(+52.2-92.3)+(-109)=-68.9\,\text{kJ mol$^{-1}$}\].
Referenced by:
No backlinks found.