combining uncertainties
Combining uncertainties (addition)
Suppose we wish to obtain the value of a physical quantity x by measuring two other quantites y and z.
If the relation between x, y and z is \[x=y+z\] or \[x=y-z\], then \[\Delta x=\Delta y+\Delta z\], where \[\Delta\] represents the uncertainty of the measurement
Example:
A liquid at 20.0±0.2C is heated to 21.5±0.5C, thus the total change in temperature is \[(21.5-20.0)\pm(0.5+0.2)\]
Combining uncertainties (fractional)
Suppose relation between x, y and z is \[x=Ayz\], where \[A\] is a constant, then \[\frac{\Delta x}{x}=\frac{\Delta y}{y}+\frac{\Delta z}{z}\].
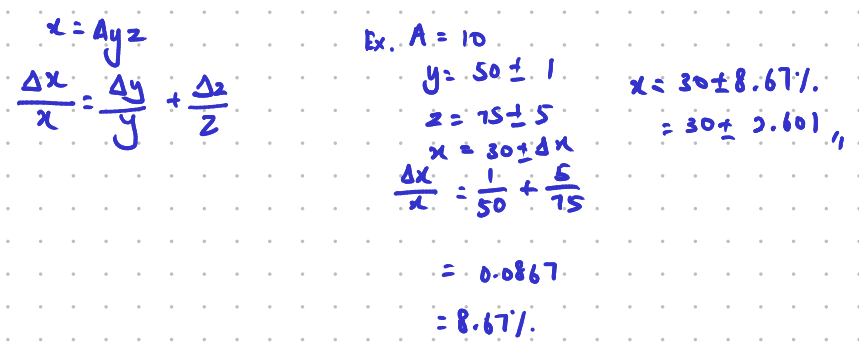
Now suppose the relation between x, y, z is \[x=Ay^{a}z^{b}\]. We're now trying to find the small changes in x, or Δx. This can be achieved by \[\frac{\Delta x}{x}=a\frac{\Delta y}{y}+b\frac{\Delta z}{z}\].
This formula can be proven as such:
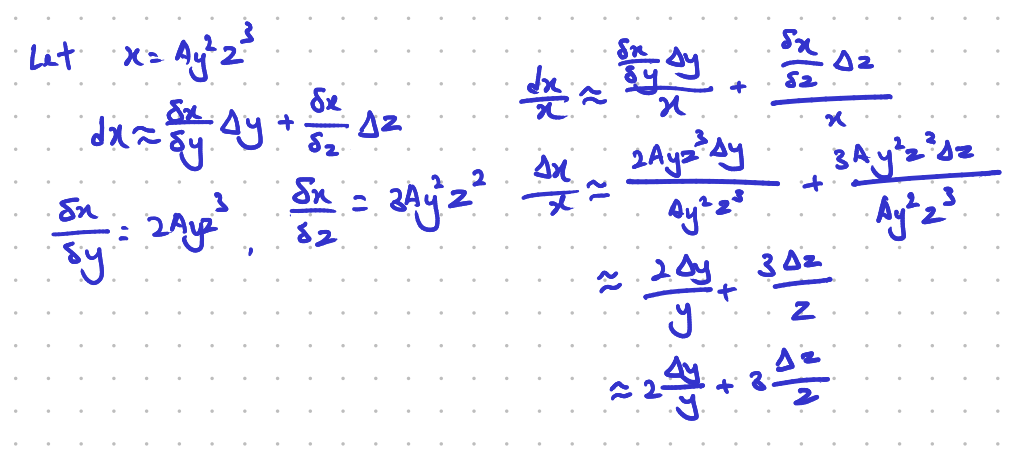
- \[dx\] = full infinitessimal change in \[x\], \[\Delta x\] is the practical (finite) changes in \[x\], while the \[\delta x\] represents partial derivative.
- To understand what \[dx\] (the full infinitesimal change) is, we have to know when we have a function \[f(y,z), df=\frac{\delta f}{\delta y}dy+\frac{\delta f}{\delta z}dz\]. Therefore \[dx\] here essetially describes the total small change of the function \[x\].
- \[dx\] and \[dy\] both represent the infinitessimally small changes in \[x\] and \[y\] while \[\Delta x\] and \[\Delta y\] represent the finite changes in \[x\] and \[y\].
- In this case we can assume that \[\Delta y\] and \[\Delta z\] are small enough to provide a good estimate of the function \[x\]'s response to changes in \[y\] and \[z\].
Referenced by:
No backlinks found.